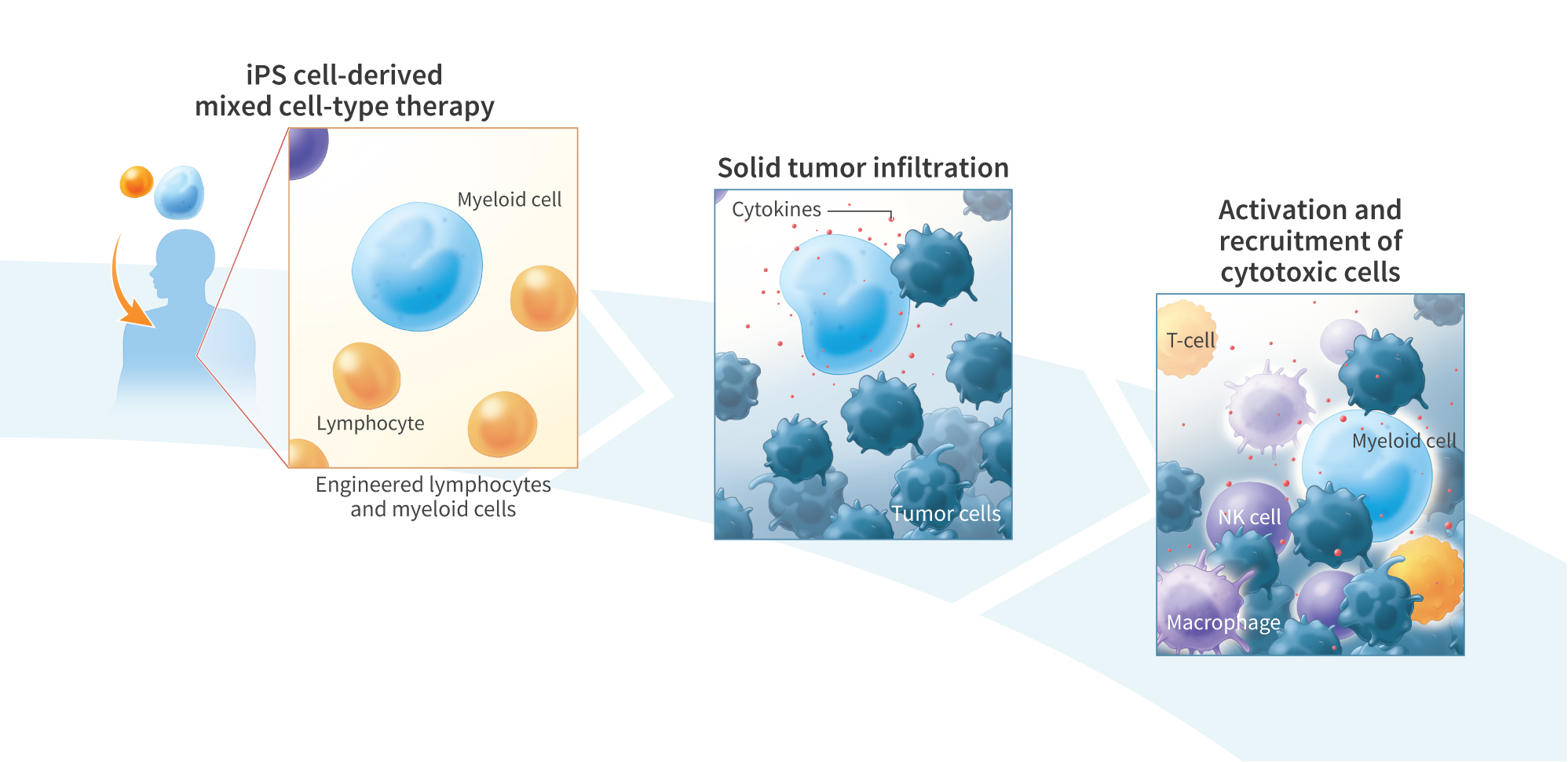
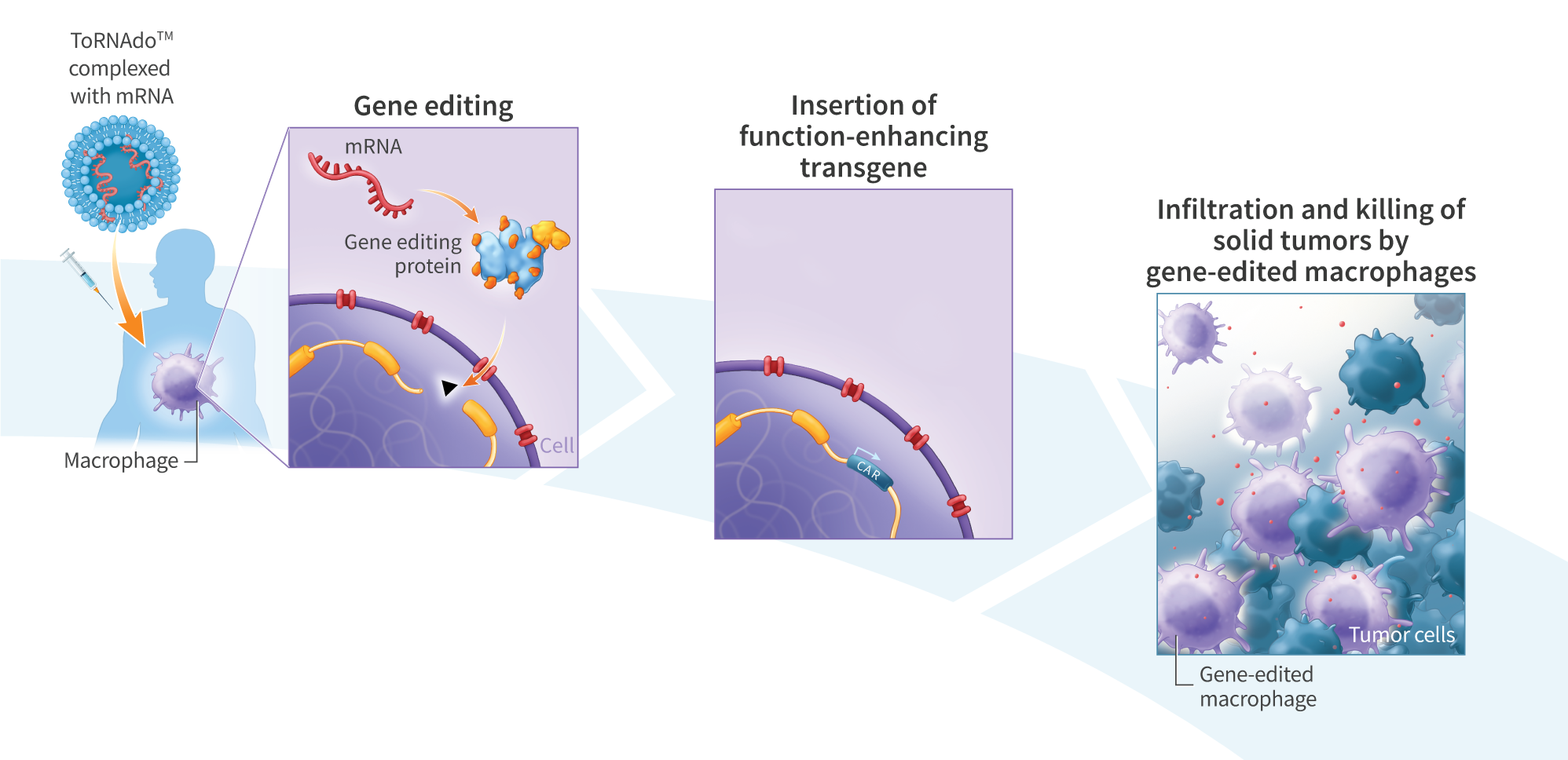
A Scalable, iPS Cell Derived Lymphocyte and Myeloid Multi-Cell-Type Therapeutic Platform for Enhanced Tumor Cell Killing
Induced pluripotent stem (iPS) cell therapies have the potential to treat a wide variety of devastating diseases. iPS$ cell-derived lymphocytes (e.g., T cells and NK cells) engineered to express targeting molecules such as chimeric antigen receptors (CARs) have shown clinical promise to treat hematological malignancies. More recently, iPS cell-derived myeloid cells are being developed to treat both hematological malignancies and solid tumors due to the ability of these cells to infiltrate and modulate the tumor microenvironment. Despite preliminary success, several challenges still remain, including poor infiltration of cytotoxic lymphocytes into solid tumors and insufficient cytotoxicity of myeloid cells. We hypothesized that a multi-cell-type therapy comprising both lymphocyte and myeloid cells may work synergistically, enhancing cytotoxicity and efficacy. We previously demonstrated the directed differentiation of mRNA reprogrammed iPS$ cells into functional cytotoxic lymphocytes and monocyte-derived M1 and M2 polarized macrophages. Here we describe a scalable bioreactor-based process for parallel differentiation of mRNA reprogrammed iPS$ cells into both CD14+ (>95% positive) macrophages and CD56bright/CD16dim NK cells. This process yields 1×10″6 myeloid cells/ml and 3×10″5 lymphoid cells/ml, and is amenable to scaling to clinically relevant doses. In vitro, the lymphoid and myeloid cells showed synergistic tumor cell killing of SKOV3 ovarian cancer cells (combined: 15.6%; macrophage alone = 2.2% (p<0.01); NK alone = 7.5% (p<0.05); E:T = 5:1). The combined cells showed increased expression of TNF-a, and demonstrated enhanced clustering and tumor cell engagement. To further improve the macrophages’ ability to target and infiltrate solid tumors, we transfected macrophages with mRNA encoding a humanized ROR1-CAR protein. mRNA transfection increased cytotoxicity towards SKOV3 cells by 6-fold. Here we describe a scalable platform for generating iPS cell-derived multi-cell-type therapies comprising both lymphoid and myeloid cells. We demonstrate that, much like the natural cellular immune response, these cells act synergistically to kill tumor cells in vitro. By more closely mimicking natural cellular immunity, multi-cell-type cell therapies represent a new class of cell therapies that may play an important role in the development of new medicines to treat cancer.