Gene-Editing Therapies for Epidermolysis Bullosa (EB)
Download our Technology Catalog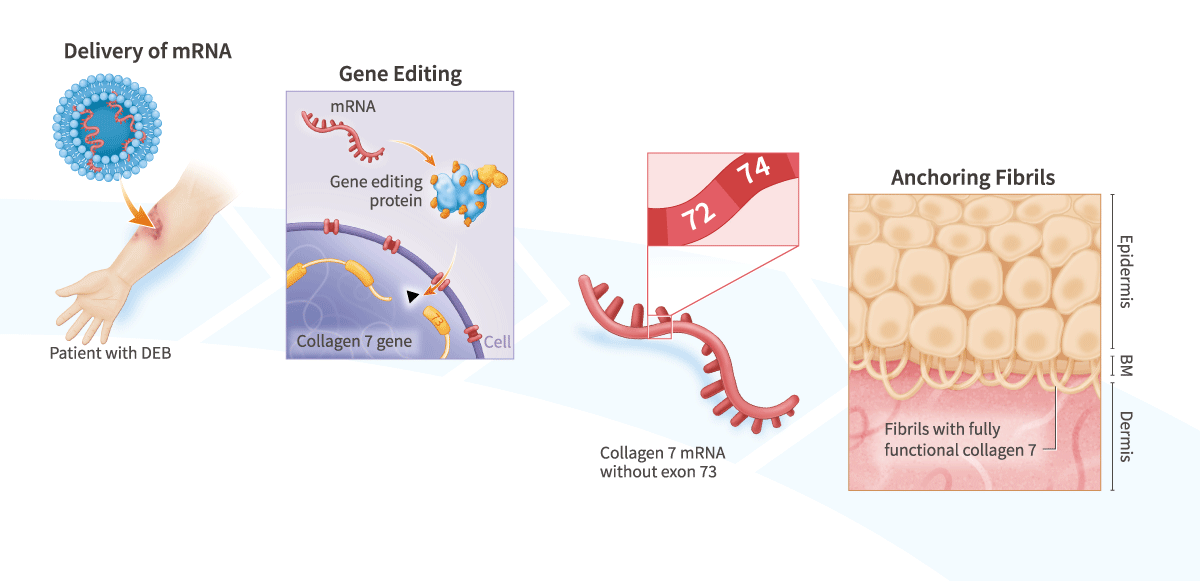
Overview
Epidermolysis Bullosa (EB) is a collection of genetic diseases caused by mutations in genes that are important for normal skin function. Many EB patients have extremely delicate skin, and in the most severe cases, live with painful blisters and open wounds over large areas of their bodies.
Our scientists developed a method for treating epidermolysis bullosa by using mRNA to express gene-editing proteins directly in a patient’s skin.
Gene-Editing Therapies for Epidermolysis Bullosa (EB) is protected by two U.S. patents (with additional patents pending in the U.S. and in other countries). Of note, certain granted patents include claims that are not limited by specific target sequence, mRNA sequence or chemistry, or type of gene-editing protein.
Example Applications
- Repair individual mutations or alter collagen 7 mRNA splicing, e.g., ablate the exon 73 splice acceptor site to generate functional collagen 7 in patients with a disease-causing mutation in exon 736
- Deliver the therapy directly to the patient’s skin – avoid ex vivo cell manipulation and skin grafts
- Combine with Factor’s Chromatin Context-Sensitive Gene-Editing Endonuclease for high-specificity in vivo gene editing
- Combine with Factor’s ToRNAdo™ Nucleic-Acid Delivery System for high efficiency in vivo delivery – proven delivery to human skin in vivo
6Mealmaker, C., et al. Mol Ther, Vol 28 No 4S1, 2020
Data
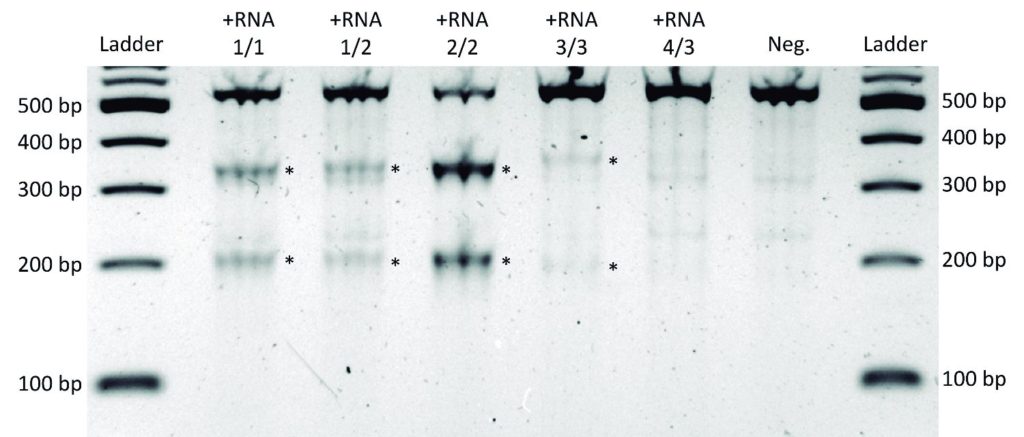
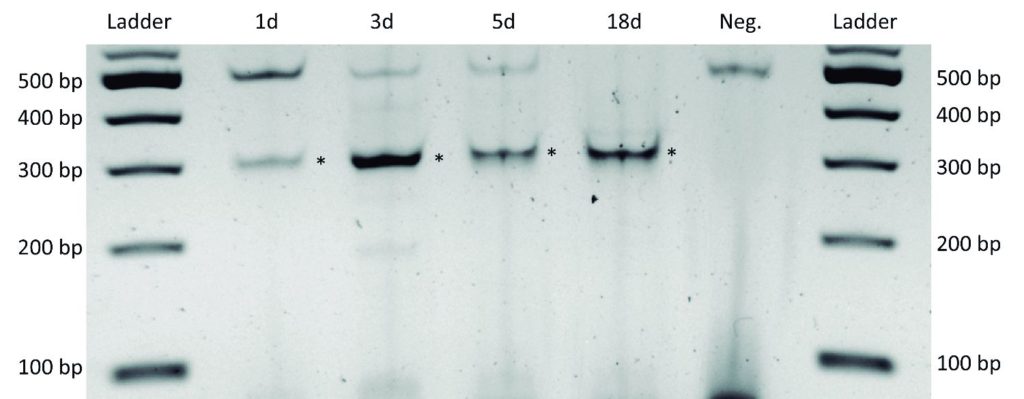
Representative Claim
U.S. Pat. No. 10,124,042
An in vivo method for treating epidermolysis bullosa, comprising
delivering a synthetic RNA encoding a gene-editing protein that targets a COL7 gene to a patient in need thereof and
inducing a single-strand or double-strand break in the COL7 gene of the patient’s keratinocytes,
thereby eliminating a mutation that is at least partially responsible for a disease phenotype, wherein:
the synthetic RNA is delivered to the patient’s keratinocytes by injection to the epidermis and
the gene-editing protein comprises a DNA-binding domain and a nuclease domain.